Proceed Ventral Patch Ifu - happyarchive. PROCEED Ventral Patch is a sterile, self- expanding, partially absorbable, flexible laminate mesh device designed for the repair of hernias and other fascial. For the Ethicon, Inc. It is indicated. MAUDE Adverse Event Report: ETHICON PROCEED VENTRAL PATCH PROCEED VENTRAL MESH PATCH.

FDA Home; Medical Devices; Databases - 5. De. Novo. PROCEED Surgical Mesh. PROCEED Surgical Mesh is a sterile, thin, flexible laminate mesh designed for the repair of hernias and other fascial deficiencies. Proceed Surgical Mesh is a hernia patch recall that was recalled by the FDA in December 2. Contact our Pensacola, FL defective product. PVP Device. PROCEED Ventral Patch PVP is a sterile, self- expanding, partially absorbable, flexible laminate mesh device designed for the repair of hernias and.
Features of the Ethicon PROCEED Ventral Patch: PVP is the first and only umbilical hernia repair device featuring lighter-weight mesh and exclusive absorb-able deployment technology; Contours with the body for secure placement. New System Capitalizes on Innovative Materials and Technology. SOMERVILLE, N.J., Dec. 16 /PRNewswire/ -- ETHICON, INC, a global medical device company, today announced the launch of the PROCEED Ventral Patch (PVP Device), the.
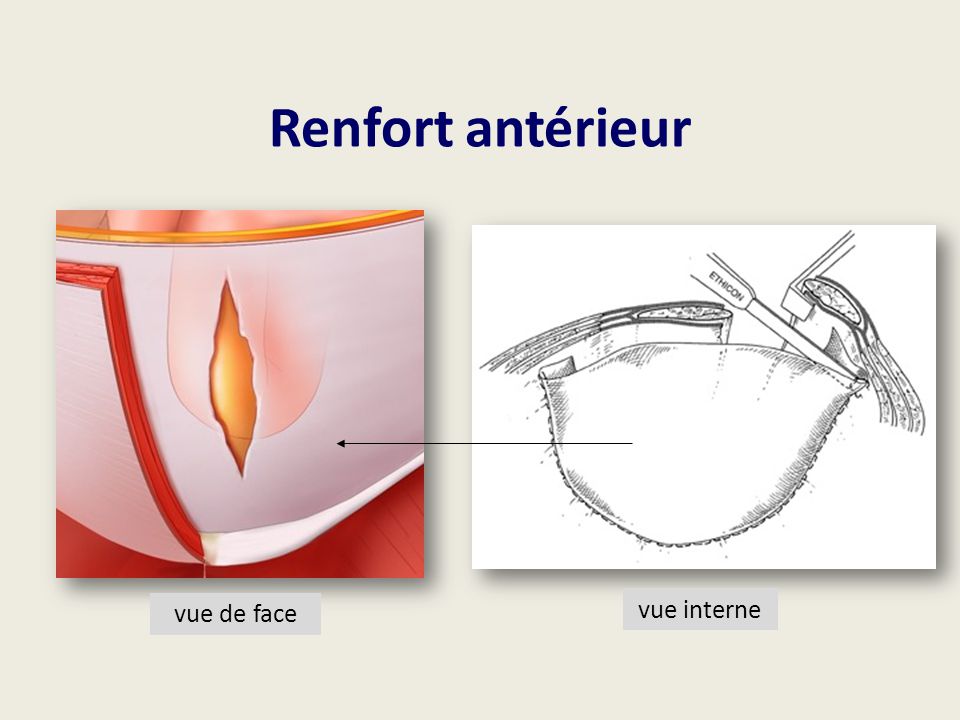
ETHICON, a Johnson & Johnson subsidiary out of Somerville, New Jersey, is making a new patch available for ventral herniorrhaphies. PVP Device is the firs. Self-Expanding Multi-Layered Partially Resorbable Lightweight Polypropylene Mesh Device (Proceed Ventral Patch CONCLUSION The Proceed Ventral Patch seems to be a good device for the treatment of small umbilical.
View This Abstract Online; Single centre observational study to evaluate the safety and efficacy of the Proceed. 2014; 18(5):671-80 (ISSN: 1248-9204) Bontinck J; Kyle. Find product information on the PROCEED Ventral Patch for hernia repair from Ethicon.

 RSS Feed
RSS Feed
